 
Therefore, the therapeutic area, eligibility criteria, treatment requirements, site structure, and staff capabilities are among the basic characteristics that sponsors and CRO must consider when evaluating sites. Estimation of the possibility of efficient use of the electronic systems available at the center (for example, electronic source documentation, electronic patient registration system, electronic patient database) Ability of the site to provide study rooms/equipment/required study procedures, including proper and regular equipment calibration and maintenance Ĥ. Qualified site staff and sufficient personnel to fulfill the study needs, including sub-Investigators, study coordinators, data managers ģ. Estimation of the capabilities of the site for recruiting patients (possible diseases, accessible populations, previous experience in recruitment, the ability to involve the patients from external sources) Ģ. The essential factors in getting the right sites for the study:ġ. Of course, CROs and sponsors are looking to get the most eligible and right sites and investigators, and this creates competition between them. The assessment may include an on-site evaluation of the facility and assessing whether the investigator and site staff are able to conduct the study in accordance with the protocol and applicable regulations. When talking about the site selection, this means the procedure of assessment and selection of potential investigators for a clinical study. As clinical development becomes more complex, an issue and consequent re-work in even one aspect of site activation can delay an entire study.Īs most part of clinical trials is global and the site selection is become multi-factorial, thus the process of site identification and forming the final site list can take a lengthy 2-3 months, on average. Even though speed and efficiency are keys, it is equally important to ensure that the project team managing the study has enough time to confirm that basic start-up factors are ready (for example, no issues with import/export, assurance that the study will be conducted in accordance with the law and there are no local restrictions for vendors setup and functioning, the electronic systems work correctly and uninterruptedly). One of the examples is too fast diving into start-up, which may occur as response to pressures due to several factors - to get to market quickly, sponsor or senior management demands, actual patient needs or competitive products or trials coming to market. Failures and incorrect actions during study start-up can impact on the overall success. Despite the impressive size of the clinical trial market, the site activation process faces several challenges. The clinical trials industry is continue steadily to grow as of January, 2020 it was found that about 52,300 of recruiting studies posted on. Each step has multiple components and requires utmost care and attention to minimize the likelihood of study delivery delays from the beginning. SSU covers a lot of activities starting from sites identification and completing the enrollment of the first patient - in between this includes site selection and pre-study visits, trial documents submission to competent authorities, contract and budget execution, vendors approval and setup, site activation, site initiation visits and, at last, enrolling the first patient. Initially, the emphasis was concentrated on conducting the study, with little attention paid to study start-ups (SSUs), but as stakeholders become more and more aware that the effectiveness of SSU processes is directly related to shorter clinical periods, interest in this opportunity is growing. Start-up phase is the base of any project at any type of business, and CROs are not an exception. 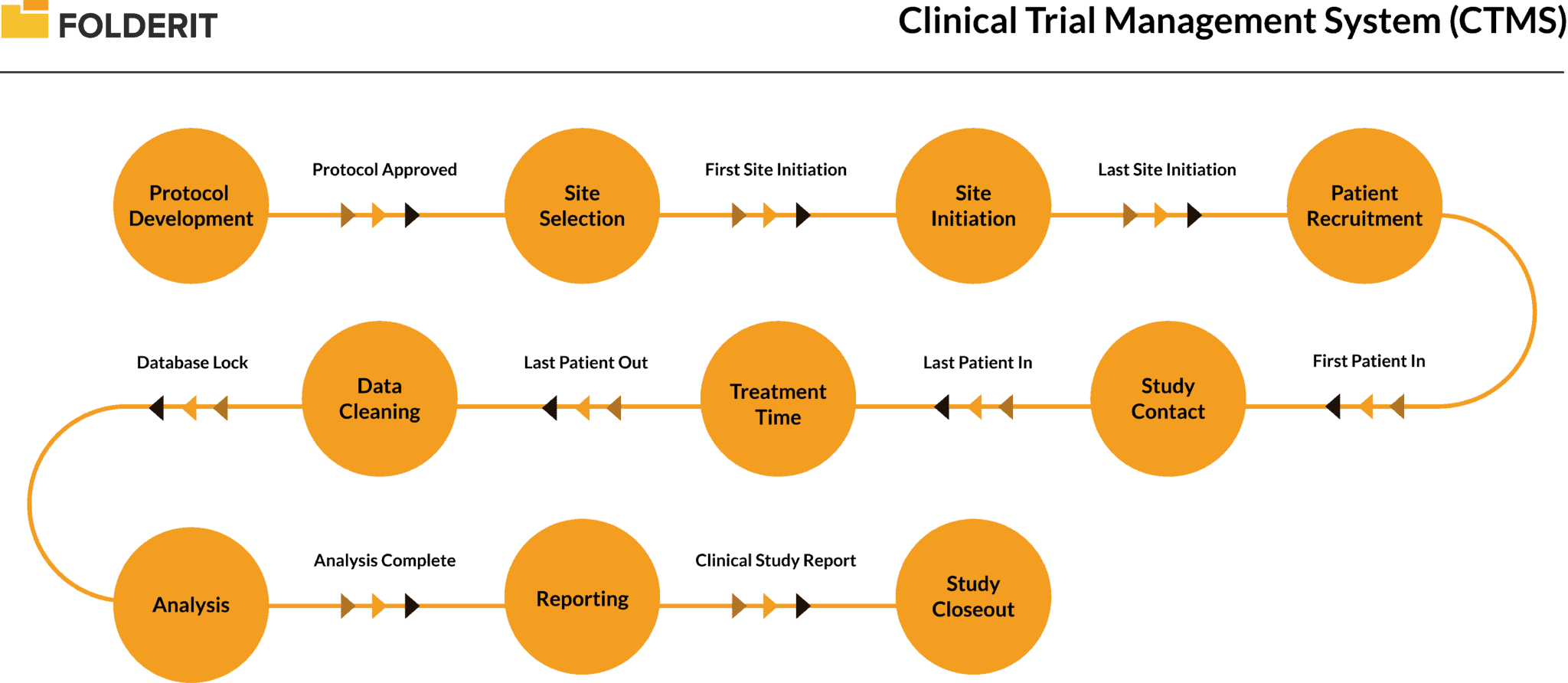
KeywordsĬlinical Trials Clinical Research Organization IWRS Introduction 
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. *Corresponding Author : Olena Barabash, Regulatory and Start-Up Manager, Global Site Activation department, Ukraine.Ĭopyright© 2020 by Barabash O. Regulatory and Start-Up Manager, Global Site Activation department, Ukraine 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
